Mitral Ballon Valvuloplasty
The percutaneous treatment of symptomatic mitral stenosis was introduced to Queensland in 1990, by Dr Aroney, who has since performed over 300 procedures. He performs all his procedures at the Holy Spirit Northside Hospital, under local anaesthetic with sedation, through a puncture in the groin.
It has become the dominant primary mode of therapy for patients with symptomatic severe mitral stenosis, and is suitable for patients of all ages, including pregnant patients.
It is used for relief of symptoms (breathlessness) in patients with severe mitral stenosis.
Indication:
Symptomatic severe mitral stenosis
Pre-procedural Assessment:

Trans-thoracic echocardiography is performed with particular attention to the mitral valve anatomy, commissural fusion and calcification, subvalvar disease, and mitral and tricuspid regurgitation.


Procedure:
It is performed under local anaesthesia, using sedation with midazolam. The left femoral artery is used for passage of a catheter to the left ventricle for gradient measurements and pressure monitoring. The right femoral vein is used for trans-septal puncture and entry of a dilator and then a catheter into the left atrium. An Inoue balloon is passed to the left atrium, and the mitral valve is crossed anterogradely. Step-wise dilatations of the mitral valve are performed with the Inous balloon, and the gradient and any mitral regurgitation are assessed haemodynamically and with trans-thoracic echocardiography after each incremental dilatation, until a satisfactory reduction in mitral gradient is obtained or there is a significant increase in mitral regurgitation. Protamine is administered and the balloon and cardiac catheters are removed.

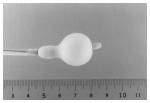


Post-procedure:
The patient is mobilised the following day when a trans-thoracic echocardiogram is performed. Injected heparin (Clexane) and warfarin are recommenced if the patient had previously been anticoagulated, and this will be required until the INR is greater than 2.0 (usually 3-4 days).
Results and complications:
- 90% of patients will have significant clinical improvement with at least a 50% improvement in mitral valve area, without severe mitral regurgitation
- 10% of patients, particularly those with heavily calcified valves, may not respond to balloon dilatation or develop significant mitral regurgitation, and may require mitral valve surgery
- Severe complications such as cardiac tamponade, need for emergency surgery, myocardial infarction or stroke are very rare in our hands (<1%).
Long-term follow-up:
- Regular clinical and echocardiographic assessment is usually performed annually, to evaluate restenosis or mitral regurgitation.
- Successful balloon valvuloplasty can result in relief of symptoms for 10 years or more.
- Repeat valvuloplasty may be performed repeatedly, if clinically required, provided severe mitral regurgitation has not developed.
Further information for referring doctors:
Absolute Contraindications:
- Severe mitral regurgitation
- Large left atrial thrombus
- Inaccessible femoral venous access
- Severe calcification of mitral commissures
- Severe clotting deficiency


Relative Contraindications (patients unsuitable or high risk for open heart surgery may be considered):
- Severe tricuspid regurgitation
- Severe eccentric calcification of mitral leaflets
- Other severe valvular disease or severe coronary disease
- Small laminated left atrial appendage thrombus
- Recent stroke
- Other severe comorbidities
If you have any further questions, please contact us at:
Queensland Cardiology
St Vincent’s Private Hospital Northside
North Medical Suites, Green Lifts Level 3,
627 Rode Road
Chermside Q 4032
(07) 3861 5522
